Advertisements
Advertisements
प्रश्न
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
उत्तर
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
Explanation:
Aldehydes and ketones are reduced to the corresponding alcohols by addition of hydrogen in the presence of catalysts (catalytic hydrogenation. It is also prepared by treating aldehydes and ketones with sodium borohydride \[\ce{(NaBH4)}\] or lithium aluminium hydride \[\ce{(LiAlH4)}\].
APPEARS IN
संबंधित प्रश्न
Write the structures of A, B, C, D and E in the following reactions:
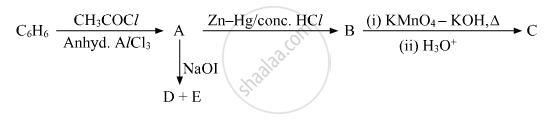
Write the reaction involved in the following:
Friedal-Crafts Alkylation of Phenol
In the reaction of phenol with CHCl3 and aqueous NaOH at 343 K, the electrophile attacking the ring is:

\[\ce{C2H5OH + SOCl2 ->[Pyridine] C2H5Cl + SO2 + HCl}\]
The above reaction is known as:
Which of the following reactions is used to prepare salicylaldehyde?
Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
Write the equations for the following reaction:
Phenol is treated with chloroform in the presence of NaOH
Why ortho-nitrophenol is steam volatile while para-nitrophenol is not?
