Advertisements
Advertisements
प्रश्न
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
उत्तर
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
Explanation:
Aldehydes and ketones are reduced to the corresponding alcohols by addition of hydrogen in the presence of catalysts (catalytic hydrogenation. It is also prepared by treating aldehydes and ketones with sodium borohydride \[\ce{(NaBH4)}\] or lithium aluminium hydride \[\ce{(LiAlH4)}\].
APPEARS IN
संबंधित प्रश्न
Write the structures of A, B, C, D and E in the following reactions:
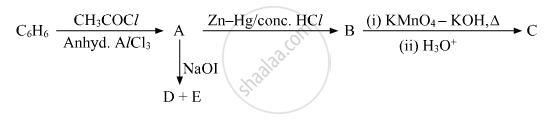
Give the equation of the following reaction:
Bromine in CS2 with phenol.
When phenol is treated with excess bromine water, it gives:
When Phenol is distilled with zinc dust, it gives:
The electrophile involved in Reimer-Tiemann reaction of phenol with CHCl3 in presence of NaOH:
Which of the following reactions is used to prepare salicylaldehyde?
Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
Write the chemical equation involved in the following reactions:
Acetylation of salicylic add
Write the name of the reaction, structure and IUPAC name of the product formed when:
Phenol reacts with CHCl3 in the presence of NaOH followed by hydrolysis.
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
