Advertisements
Advertisements
Question
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
Solution
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
Explanation:
Aldehydes and ketones are reduced to the corresponding alcohols by addition of hydrogen in the presence of catalysts (catalytic hydrogenation. It is also prepared by treating aldehydes and ketones with sodium borohydride \[\ce{(NaBH4)}\] or lithium aluminium hydride \[\ce{(LiAlH4)}\].
APPEARS IN
RELATED QUESTIONS
Write the final product(s) in each of the following reactions:
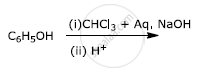
Name the reagent used in the following reaction:
Bromination of phenol to 2, 4, 6-tribromophenol.
Explain the following with an example.
Kolbe’s reaction.
Write the reaction involved in the following:
Friedal-Crafts Alkylation of Phenol
On distilling phenol with Zn dust, one gets:
The electrophile involved in Reimer-Tiemann reaction of phenol with CHCl3 in presence of NaOH:
Write the equations for the following reaction:
Phenol is treated with chloroform in the presence of NaOH
Why ortho-nitrophenol is steam volatile while para-nitrophenol is not?
For the pair phenol and cyclohexanol, answer the following:
Give one chemical test to distinguish between the two.
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
