Advertisements
Advertisements
प्रश्न
Why ortho-nitrophenol is steam volatile while para-nitrophenol is not?
उत्तर
In o-nitrophenol, oxygen in the NO2 group forms an intramolecular hydrogen bond with hydrogen in the hydroxide group. While in the p-nitrophenol, oxygen in the NO2 group of one molecule forms an intermolecular hydrogen bond with hydrogen in the hydroxide group of another molecule.
Since intramolecular hydrogen bonds are weaker than intermolecular hydrogen bonds, o-nitrophenol is less steam volatile than p-nitrophenol.
APPEARS IN
संबंधित प्रश्न
Give simple chemical tests to distinguish between the following pairs of compounds: Benzoic acid and Phenol
Write the main products when
2, 4, 6-trinitrochlorobenzene is subjected to hydrolysis
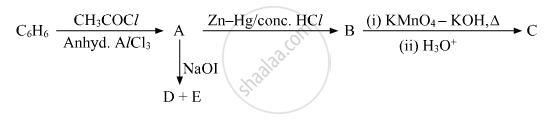
Write the structures of A, B, C, D and E in the following reactions:
While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
Name the reagent used in the following reaction:
Bromination of phenol to 2, 4, 6-tribromophenol.
Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 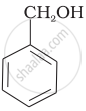 |
| (C) | 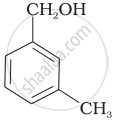 |
| (D) | 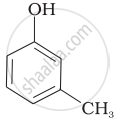 |
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
Which of the following reacts with phenol to give salicylaldehyde after hydrolysis?
