Advertisements
Advertisements
प्रश्न
Write the main products when
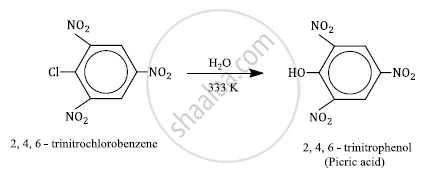
2, 4, 6-trinitrochlorobenzene is subjected to hydrolysis
उत्तर
2, 4, 6-trinitrochlorobenzene under mild hydrolysis conditions (H2O/323 K) gives 2, 4, 6-trinitrophenol or picric acid.
APPEARS IN
संबंधित प्रश्न
Write the main product(s) in each of the following reactions:
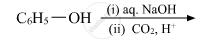
Give the equation of the following reaction:
Bromine in CS2 with phenol.
Name the reagent used in the following reaction:
Bromination of phenol to 2, 4, 6-tribromophenol.
Picric acid is ____________.
When phenol is treated with excess bromine water, it gives:
When phenol is heated with CHCl3 and alcoholic KOH when salicylaldehyde is produced. This reaction is known as ____________.
Which of the following species can act as the strongest base?
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) |  |
| (C) |  |
| (D) | 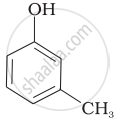 |
Convert the following:
Phenol to N-phenylethanamide.
Which of the following is not aromatic?
