Advertisements
Advertisements
प्रश्न
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 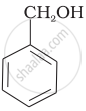 |
| (C) | 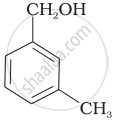 |
| (D) | 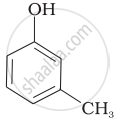 |
विकल्प
A, B, C, D
A, D
B, C
A
उत्तर
B, C
Explanation:
Compound (A) i.e., phenol and compound (D) i.e., a derivative of phenol cannot be considered as aromatic alcohol. As phenol is also known as, carbolic acid cannot be considered as aromatic alcohol.
Compound (B) and (C), –OH group is bonded to sp3 hybridised carbon which in turn is bonded to benzene ring.
APPEARS IN
संबंधित प्रश्न
Write the main products when
2, 4, 6-trinitrochlorobenzene is subjected to hydrolysis
Write the structures of A, B, C, D and E in the following reactions:
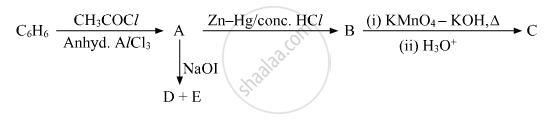
Name the reagent used in the following reaction:
Bromination of phenol to 2, 4, 6-tribromophenol.
Explain the following with an example.
Kolbe’s reaction.
Picric acid is ____________.
On distilling phenol with Zn dust, one gets:
Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Attacking species in nitration of benzene in presence of fuming HNO3 is
Write the chemical equation involved in the following reactions:
Acetylation of salicylic add
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
