Advertisements
Advertisements
Question
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 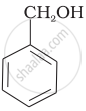 |
| (C) | 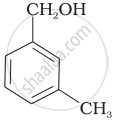 |
| (D) | 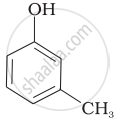 |
Options
A, B, C, D
A, D
B, C
A
Solution
B, C
Explanation:
Compound (A) i.e., phenol and compound (D) i.e., a derivative of phenol cannot be considered as aromatic alcohol. As phenol is also known as, carbolic acid cannot be considered as aromatic alcohol.
Compound (B) and (C), –OH group is bonded to sp3 hybridised carbon which in turn is bonded to benzene ring.
APPEARS IN
RELATED QUESTIONS
Write the structures of A, B, C, D and E in the following reactions:
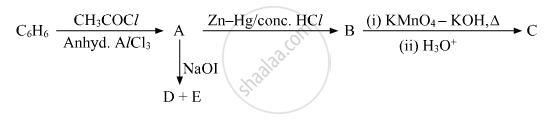
Write the equation involved in the following reaction:
Kolbe’s reaction
While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
Give the equation of the following reaction:
Bromine in CS2 with phenol.
Explain the following with an example.
Kolbe’s reaction.
Picric acid is ____________.
When phenol is treated with excess bromine water, it gives:
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
Why ortho-nitrophenol is steam volatile while para-nitrophenol is not?
For the pair phenol and cyclohexanol, answer the following:
Give one chemical test to distinguish between the two.
