Advertisements
Advertisements
Questions
Write the equation involved in the following reaction:
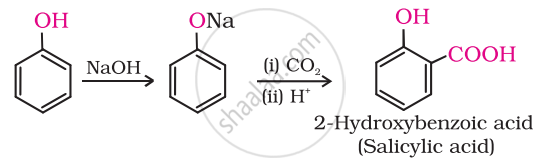
Kolbe’s reaction
Explain the following with an example.
Kolbe’s reaction
Solution
Phenoxide ion generated by treating phenol with sodium hydroxide is even more reactive than phenol towards electrophilic aromatic substitution. Hence, it undergoes electrophilic substitution with carbon dioxide, a weak electrophile. Ortho hydroxybenzoic acid is formed as the main reaction product.
RELATED QUESTIONS
Give simple chemical tests to distinguish between the following pairs of compounds: Benzoic acid and Phenol
Write the main products when
2, 4, 6-trinitrochlorobenzene is subjected to hydrolysis
Write the main product(s) in each of the following reactions:
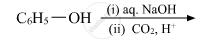
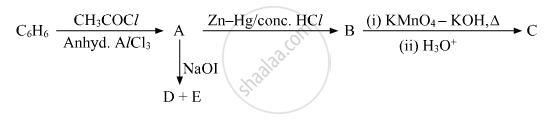
Write the structures of A, B, C, D and E in the following reactions:
Write the equation involved in the following reaction:
Reimer-Tiemann reaction
Name the reagent used in the following reaction:
Bromination of phenol to 2, 4, 6-tribromophenol.
Explain the following with an example.
Kolbe’s reaction.
Picric acid is ____________.
On distilling phenol with Zn dust, one gets:
The electrophile involved in Reimer-Tiemann reaction of phenol with CHCl3 in presence of NaOH:
Which of the following reactions is used to prepare salicylaldehyde?
Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 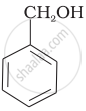 |
| (C) | 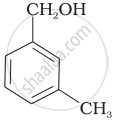 |
| (D) | 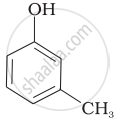 |
Which of the following are benzylic alcohols?
(i) \[\ce{C6H5 - CH2 - CH2OH}\]
(ii) \[\ce{C6H5 - CH2OH}\]
(iii) \[\begin{array}{cc}
\ce{C6H5 - CH - OH}\\
\phantom{}|\phantom{.}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\]
(iv) \[\begin{array}{cc}
\ce{C6H5 - CH2 - CH - OH}\\
\phantom{.......}|\phantom{}\\
\phantom{.........}\ce{CH3}\phantom{}
\end{array}\]
Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
Nitration is an example of aromatic electrophilic substitution and its rate depends upon the group already present in the benzene ring. Out of benzene and phenol, which one is more easily nitrated and why?
For the pair phenol and cyclohexanol, answer the following:
Give one chemical test to distinguish between the two.
Write the name of the reaction, structure and IUPAC name of the product formed when:
Phenol reacts with CHCl3 in the presence of NaOH followed by hydrolysis.
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
