Advertisements
Advertisements
प्रश्न
Write the main product(s) in each of the following reactions:
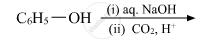
उत्तर
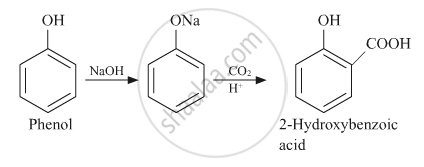
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:
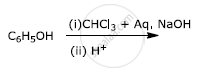
Write the main products when
2, 4, 6-trinitrochlorobenzene is subjected to hydrolysis
When phenol is treated with excess bromine water, it gives:
When phenol is heated with CHCl3 and alcoholic KOH when salicylaldehyde is produced. This reaction is known as ____________.
The electrophile involved in Reimer-Tiemann reaction of phenol with CHCl3 in presence of NaOH:
Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
Nitration is an example of aromatic electrophilic substitution and its rate depends upon the group already present in the benzene ring. Out of benzene and phenol, which one is more easily nitrated and why?
Convert the following:
Phenol to N-phenylethanamide.
Write the equations for the following reaction:
Phenol is treated with chloroform in the presence of NaOH
