Commerce (English Medium)
Science (English Medium)
Arts (English Medium)
Academic Year: 2015-2016
Date & Time: 9th March 2016, 10:30 am
Duration: 3h
Advertisements
Write the IUPAC name of the given compound:
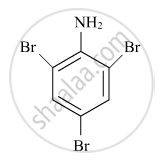
Chapter: [0.05] Coordination Compounds
What is the reason for the stability of colloidal sols?
Chapter: [0.05] Surface Chemistry
Out of 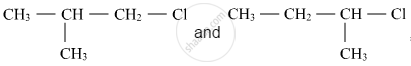 , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Chapter: [0.06] Haloalkanes and Haloarenes
On adding NaOH to ammonium sulphate, a colourless gas with pungent odour is evolved, which forms a blue-coloured complex with Cu2+ ion. Identify the gas.
Chapter: [0.05] Coordination Compounds
What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction?
Chapter: [0.01] Solid State
For a reaction : 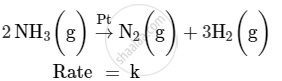
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
Chapter: [0.03] Chemical Kinetics
Write the mechanism of the following reaction:
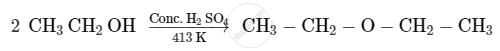
Chapter: [0.07] Alcohols, Phenols and Ethers
From the given cells:
Lead storage cell, Mercury cell, Fuel cell and Dry cell
Answer the following:
(i) Which cell is used in hearing aids?
(ii) Which cell was used in Apollo Space Programme?
(iii) Which cell is used in automobiles and inverters?
(iv) Which cell does not have long life?
Chapter: [0.02] Electrochemistry
When chromite ore FeCr2O4 is fused with NaOH in presence of air, a yellow-coloured compound (A) is obtained, which on acidification with dilute sulphuric acid gives a compound (B). Compound (B) on reaction with KCl forms an orange coloured crystalline compound (C).
(i) Write the formulae of the compounds (A), (B) and C.
(ii) Write one use of compound (C).
Chapter: [0.04] d-block and f-block Elements
Complete the following chemical equation
8MnO4- + 3S2O32- + H2O →
Chapter: [0.04] d-block and f-block Elements
Complete the following chemical equation
Cr2O72- + 3Sn2+ + 14H+ →
Chapter: [0.04] d-block and f-block Elements
When a co-ordination compound CrCl3.6H2O is mixed with AgNO3, 2 moles of AgCl are precipitated per mole of the compound. Write
(i) Structural formula of the complex.
(ii) IUPAC name of the complex.
Chapter: [0.05] Coordination Compounds
Differentiate between adsorbtion and absorption.
Chapter: [0.05] Surface Chemistry
Out of MgCl2 and AlCl3, which one is more effective in causing coagulation of negatively charged sol and why?
Chapter: [0.05] Surface Chemistry
Out of sulphur sol and proteins, which one form multimolecular colloids?
Chapter: [0.05] Surface Chemistry
Name the method of refining of metals such as Germanium.
Chapter: [0.06] General Principles and Processes of Isolation of Elements
In the extraction of Al, impure Al2O3 is dissolved in conc. NaOH to form sodium aluminate and leaving impurities behind. What is the name of this process?
Chapter: [0.06] General Principles and Processes of Isolation of Elements
What is the role of coke in the extraction of iron from its oxides?
Chapter: [0.06] General Principles and Processes of Isolation of Elements
Calculate e.m.f of the following cell at 298 K:
2Cr(s) + 3Fe2+ (0.1M) → 2Cr3+ (0.01M) + 3 Fe(s)
Given: E°(Cr3+ | Cr) = – 0.74 VE° (Fe2+ | Fe) = – 0.44 V
Chapter: [0.02] Electrochemistry
Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Chapter: [0.06] Haloalkanes and Haloarenes
Given reasons: The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Chapter: [0.06] Haloalkanes and Haloarenes
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Chapter: [0.06] Haloalkanes and Haloarenes
Advertisements
An element crystallizes in a f.c.c. lattice with a cell edge of 250 pm. Calculate the density if 300 g of this element contains 2 × 1024 atoms.
Chapter: [0.01] Solid State
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
Chapter: [0.03] Chemical Kinetics
What is the role of t-butyl peroxide in the polymerization of ethene?
Chapter: [0.15] Polymers
Identify the monomers in the following polymer:
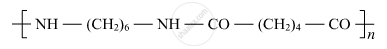
Chapter:
Arrange the following polymers in the increasing order of their intermolecular forces:
Polystyrene, Terylene, Buna-S
Chapter:
Write the mechanism of free radical polymerization of ethene.
Chapter: [0.15] Polymers
Write the name of two monosaccharides obtained on hydrolysis of lactose sugar.
Chapter: [0.1] Biomolecules
Why Vitamin C cannot be stored in our body ?
Chapter: [0.1] Biomolecules
What is the difference between a nucleoside and nucleotide?
Chapter: [0.1] Biomolecules
For the complex [Fe(CN)6]3–, write the hybridization type, magnetic character and spin nature of the complex. (At. number : Fe = 26).
Chapter: [0.05] Coordination Compounds
Draw one of the geometrical isomers of the complex [Pt(en)2Cl2]2+ which is optically active.
Chapter: [0.05] Coordination Compounds
Account for the following:
Mn shows the highest oxidation state of +7 with oxygen but with fluorine, it shows oxidation state of +4.
Chapter: [0.04] d-block and f-block Elements
Give reasons: Transition metals show variable oxidation states.
Chapter: [0.04] d-block and f-block Elements
Give a reason for the following:
Actinoids show irregularities in their electronic configurations.
Chapter: [0.04] d-block and f-block Elements
Write the main product(s) in each of the following reactions:
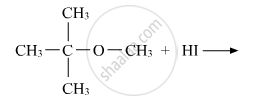
Chapter: [0.07] Alcohols, Phenols and Ethers
Write the main product(s) in each of the following reactions:
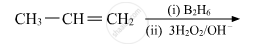
Chapter: [0.07] Alcohols, Phenols and Ethers
Write the main product(s) in each of the following reactions:
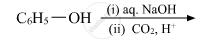
Chapter: [0.07] Alcohols, Phenols and Ethers
Write the structures of A, B and C in the following:
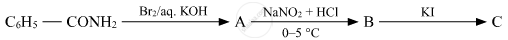
Chapter: [0.09] Amines
Write the structures of A, B and C in the following:

Chapter: [0.06] Haloalkanes and Haloarenes
Advertisements
Due to hectic and busy schedule, Mr Angad made his life full of tensions and anxiety. He started taking sleeping pills to overcome the depression without consulting the doctor. Mr Deepak, a close friend of Mr. Angad advised him to stop taking sleeping pills and suggested to change his life lifestyle by doing yoga, meditation and some physical exercise. Mr. Angad followed his friend’s advice and after few days he started feeling better.
After reading the above passage, answer the following
(i) What are the values (at least two) displayed by Mr. Deepak?
(ii) Why is it not advisable to take sleeping pills without consulting doctor?
(iii) What are tranquilisers? Give two examples.
Chapter: [0.16] Chemistry in Everyday Life
Calculate the freezing point of solution when 1.9 g of MgCl2 (M = 95 g mol−1) was dissolved in 50 g of water, assuming MgCl2 undergoes complete ionization.
(Kf for water = 1.86 K kg mol−1)
Chapter: [0.01] Solutions
Out of 1 M glucose and 2 M glucose, which one has a higher boiling point and why?
Chapter: [0.01] Solutions
What happens when the external pressure applied becomes more than the osmotic pressure of solution?
Chapter: [0.01] Solutions
When 2.56 g of sulphur was dissolved in 100 g of CS2, the freezing point lowered by 0.383 K. Calculate the formula of sulphur (Sx).
(Kf for CS2 = 3.83 K kg mol−1, Atomic mass of sulphur = 32 g mol−1]
Chapter: [0.01] Solutions
Blood cells are isotonic with 0.9% sodium chloride solution. What happens if we place blood cells in a solution containing
(i) 1.2% sodium chloride solution?
(ii) 0.4% sodium chloride solution?
Chapter: [0.01] Solutions
Account for the following :
Ozone is thermodynamically unstable.
Chapter: [0.07] P - Block Elements
Account for the following : Solid PCl5 is ionic in nature.
Chapter: [0.07] P - Block Elements
Account for the following :
Fluorine forms only one oxoacid HOF
Chapter: [0.07] P - Block Elements
Draw the structure of BrF5
Chapter: [0.07] P - Block Elements
Draw the structures of the following molecules: XeF4
Chapter: [0.07] P - Block Elements
Compare the oxidizing action of F2 and Cl2 by considering parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
Chapter: [0.07] P - Block Elements
Write the conditions to maximize the yield of H2SO4 by contact process.
Chapter: [0.07] P - Block Elements
Arrange the following in the increasing order of property mentioned :
H3PO3, H3PO4, H3PO2 (Reducing character)
Chapter: [0.07] P - Block Elements
Arrange the following in the increasing order of property mentioned :
NH3, PH3, AsH3, SbH3, BiH3 (Base strength)
Chapter: [0.07] P - Block Elements
Write the structures of A, B, C, D and E in the following reactions:
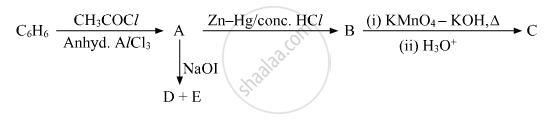
Chapter: [0.07] Alcohols, Phenols and Ethers
Write the chemical equation for the reaction involved in Cannizzaro reaction.
Chapter: [0.08] Aldehydes, Ketones and Carboxylic Acids
Draw the structure of the semicarbazone of ethanal.
Chapter: [0.08] Aldehydes, Ketones and Carboxylic Acids
Why pKa of F-CH2-COOH is lower than that of Cl−CH2−COOH?
Chapter: [0.08] Aldehydes, Ketones and Carboxylic Acids
Write the product in the following reaction:
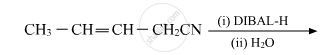
Chapter: [0.08] Aldehydes, Ketones and Carboxylic Acids
How can you distinguish between propanal and propanone?
Chapter: [0.08] Aldehydes, Ketones and Carboxylic Acids
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 12 Chemistry with solutions 2015 - 2016
Previous year Question paper for CBSE Class 12 -2016 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 12.
How CBSE Class 12 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
