Advertisements
Advertisements
प्रश्न
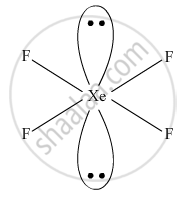
Draw the structures of the following molecules: XeF4
Write the structures of the following:
XeF4
उत्तर
XeF4, Square planar
APPEARS IN
संबंधित प्रश्न
Complete the equation : XeF2+PF5 →
Give the formula and describe the structure of a noble gas species which is isostructural with:
`IBr_2^(-)`
Write electronic configuration and two uses of neon. (Z = 10)
Fill in the blanks by choosing the appropriate word/words from the brackets
(square pyramidal, electrical, 74; 26, sp3d2, sp3d, chemical, 68, 32, tetrahedral, yellow, white, iodoform, Lucas)
The geometry of XeOF4 molecule is ______ and the hybridisation of Xenon atom in the molecule is ________.
Explain the trend in the following atomic properties of group 16 elements:
Electron gain enthalpy
Which of the following fluorides does not exist?
Match the compounds given in Column I with the hybridisation and shape given in Column II and mark the correct option.
| Column I | Column II |
| (A) XeF6 | (1) sp3d3 – distorted octahedral |
| (B) XeO3 | (2) sp3d2 – square planar |
| (C) XeOF4 | (3) sp3 – pyramidal |
| (D) XeF4 | (4) sp3d2 – square pyramidal |
The atomic radii of Zr and Hf are almost identical. This is because of
\[\ce{XeF6 + H2O ->[Partial][Hydrolysis] \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
Write two uses of neon.
