Advertisements
Advertisements
प्रश्न
Arrange the following in the increasing order of property mentioned :
H3PO3, H3PO4, H3PO2 (Reducing character)
उत्तर
(a) Reducing character: H3PO4 < H3PO3 < H3PO2
This trend can be explained on the basis of structures of oxyacids of phosphorus.
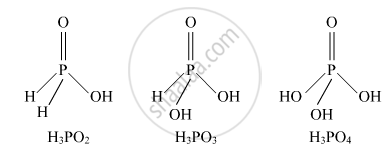
The higher the number of P−H bond, the higher the reducing power of the oxyacid of phosphorus. There are two P−H bonds in H3PO2, one P−H bond in H3PO3 and no P−H bond in H3PO4. So, this explains the reducing trend of the given oxyacids of phosphorus.
APPEARS IN
संबंधित प्रश्न
Explain how does nitrogen exhibit anomalous behaviour amongst group 15 elements.
The group 15 element having inner electronic configuration as of argon is-
(a) Phosphorous (z = 15)
(b) Antimony (z =51)
(c) Arsenic (z = 33)
(d) Nitrogen (z = 7)
Account for the following :
BiH3 is the strongest reducing agent amongst all the hydrides of group 15.
Account for the following
Bi(V) is a stronger oxidizing agent than Sb(V).
Which among the following forms basic oxide?
Which of the following is the strongest reducing agent?
The correct order of oxidising power is:
In \[\ce{PCl5}\], phosphorus is in sp3d hybridised state but all its five bonds are not equivalent. Justify your answer with reason.
Hess's law is used to calculate:
The elements which are characterized by the outer electronic configuration ns1 to ns2 np6 are collectively called
