Advertisements
Advertisements
प्रश्न
In \[\ce{PCl5}\], phosphorus is in sp3d hybridised state but all its five bonds are not equivalent. Justify your answer with reason.
उत्तर
In \[\ce{PCl5}\], phosphorus undergoes sp3d hybridisation and a trigonal bipyramidal configuration comes into existence.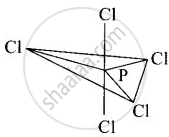
The equatorial \[\ce{P - Cl}\] bonds are equivalent while two axial bonds are different and larger than equatorial bonds.
APPEARS IN
संबंधित प्रश्न
Among the hydrides of Group-15 elements, which have the maximum reducing character?
Give reasons When Cl2 reacts with the excess of F2, ClF3 is formed and not FCl3.
[Ar]3d104s24p3 is the electronic configuration of ____________.
The tribasic acid is ____________.
The strongest oxidising agent is ____________.
The correct decreasing order of basic strength is:
If chlorine gas is passed through hot NaOH solution, two changes are observed in the oxidation number of chlorine during the reaction. These are :
(i) 0 to +5
(ii) 0 to +3
(iii) 0 to –1
(iv) 0 to +1
The elements with atomic numbers 9, 17, 35, 53 and 85 belong to
In a regular octahedral molecule mx6, the number of X – M – X bouds at 180° are:-
The basic strengths of the hydrides group is elements decrease in the order.
