Advertisements
Advertisements
प्रश्न
In \[\ce{PCl5}\], phosphorus is in sp3d hybridised state but all its five bonds are not equivalent. Justify your answer with reason.
उत्तर
In \[\ce{PCl5}\], phosphorus undergoes sp3d hybridisation and a trigonal bipyramidal configuration comes into existence.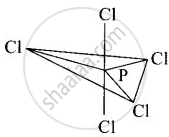
The equatorial \[\ce{P - Cl}\] bonds are equivalent while two axial bonds are different and larger than equatorial bonds.
APPEARS IN
संबंधित प्रश्न
Why is BiH3 the strongest reducing agent amongst all the hydrides of Group 15 elements?
Account for the following :
BiH3 is the strongest reducing agent amongst all the hydrides of group 15.
Account for the following
The enthalpy of atomisation is lowest for Zn in 3d series of the transition elements.
Ionic radii (in Å) of As3+, Sb3+ and Bi3+ follow the order:
Which of the following species has the highest dipole moment?
With respect to protonic acids, which of the following statement is correct?
Which of the following statements is wrong?
Which of the following statements are correct for \[\ce{SO2}\] gas?
(i) It acts as bleaching agent in moist conditions.
(ii) It’s molecule has linear geometry.
(iii) It’s dilute solution is used as disinfectant.
(iv) It can be prepared by the reaction of dilute \[\ce{H2SO4}\] with metal sulphide.
Match the items of Columns I and II and mark the correct option.
| Column I | Column II | |
| (A) | Its partial hydrolysis does not | (1) He |
| (B) | It is used in modern diving apparatus | (2) XeF6 |
| (C) | It is used to provide inert atmosphere for filling electrical bulbs |
(3) XeF4 |
| (D) | Its central atom is in sp3d2 hybridisation | (4) Ar |
Chemical nature of the nitrogen oxide compound obtained from a reaction of concentrated nitric acid and P4O10 (in 4 : 1 ratio) is ______.
