Advertisements
Advertisements
प्रश्न
Arrange the following in the increasing order of property mentioned :
H3PO3, H3PO4, H3PO2 (Reducing character)
उत्तर
(a) Reducing character: H3PO4 < H3PO3 < H3PO2
This trend can be explained on the basis of structures of oxyacids of phosphorus.
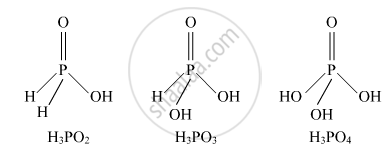
The higher the number of P−H bond, the higher the reducing power of the oxyacid of phosphorus. There are two P−H bonds in H3PO2, one P−H bond in H3PO3 and no P−H bond in H3PO4. So, this explains the reducing trend of the given oxyacids of phosphorus.
APPEARS IN
संबंधित प्रश्न
Why are pentahalides more covalent than trihalides?
Why does R3P=O exist but R3N=O does not (R = alkyl group)?
Nitrogen exists as diatomic molecule and phosphorus as P4. Why?
The oxidation state of nitrogen in dinitrogen trioxide is ____
(a) + 1
(b) + 2
(c) + 3
(d) + 4
The correct order of solubility in water for He, Ne, Ar, Kr, Xe is:
Which of the following group 15 elements forms metallic bonds in an elemental state?
Elements of group-15 form compounds in +5 oxidation state. However, bismuth forms only one well-characterised compound in +5 oxidation state. The compound is ______.
The oxidation state of central atom in the anion of compound \[\ce{NaH2PO2}\] will be ______.
The enthalpy change of a reaction does not depends upon?
The elements with zero electron affinity are
