Advertisements
Advertisements
प्रश्न
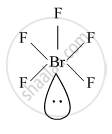
Draw the structure of BrF5
उत्तर
BrF5
APPEARS IN
संबंधित प्रश्न
Draw the structures of the following: BrF5
What is the oxidation state of bromine in the product?
\[\ce{Br2 + \underset{(excess)}{3F2} ->?}\]
The bent T-shaped interhalogen is ____________.
Which among the following is the most reactive?
Which of the following interhalogen is a pale brown gas at room temperature?
Which of the following interhalogen is a colourless liquid at room temperature?
The number of lone pair of electrons present on central atom in the interhalogen XX'3 molecule:
Which of the following is not the characteristic of interhalogen compounds?
Match the compounds given in Column I with the hybridisation and shape given in Column II and mark the correct option.
| Column I | Column II |
| (A) XeF6 | (1) sp3d3 – distorted octahedral |
| (B) XeO3 | (2) sp3d2 – square planar |
| (C) XeOF4 | (3) sp3 – pyramidal |
| (D) XeF4 | (4) sp3 d2 – square pyramidal |
Brown ring test is used for detection of which radical?
