Advertisements
Advertisements
प्रश्न
Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
उत्तर
In chlorobenzene, the C−Cl bond gets partial double bond character because of the +M effect shown by the Cl group. Due to double bond character, the bond length decreases in chlorobenzene as compared to the normal CH3−Cl bond length. Therefore, the C–Cl bond length in chlorobenzene is shorter than the C–Cl bond length in CH3–Cl.
संबंधित प्रश्न
Give reasons for the following:
(CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH.
Halogenation of alkanes is ____________.
Which of the following statements are correct about this reaction?
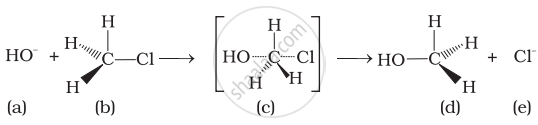
(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
CCl4 is insoluble in water because:-
Optical activity of an enantiomeric mixture is +12.6° and the specific rotation of (+) isomer is +30°. The optical purity is ______ %.
Inversion of configuration occurs in ______.
Complete the reaction with the main product formed:

