Advertisements
Advertisements
प्रश्न
Which of the following statements are correct about this reaction?
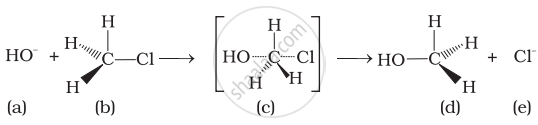
(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
उत्तर
(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
Explanation:
In the given reaction, alkyl halide is primary in nature. Here, a transitory state is observed in which one bond is broken and one bond is formed synchronously he., in one step. So, it follows SN2 mechanism.
In this mechanism, nucleophile attacks the carbon at 180° to the leaving group. So the reactant and product have opposite configuration.
APPEARS IN
संबंधित प्रश्न
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
AgCN reacts with haloalkanes to form isocyanide. Haloalkanes react with KCN to form alkyl cyanides as the main product. Why?
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
An important chemical method to resolve a racemic mixture makes use of the formation of ______.
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
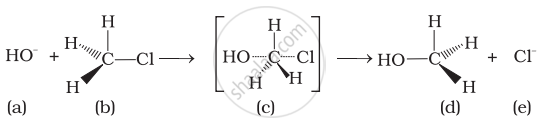
Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
In which reaction mechanism carbocation is formed?
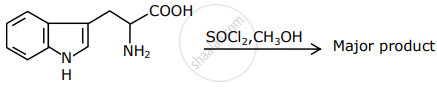
The major product formed in the following reaction is:
Optical activity of an enantiomeric mixture is +12.6° and the specific rotation of (+) isomer is +30°. The optical purity is ______ %.
Complete the reaction with the main product formed:

