Advertisements
Advertisements
प्रश्न
Which of the following statements are correct about the reaction intermediate?
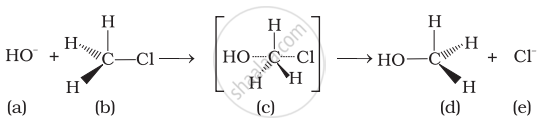
(i) Intermediate (c) is unstable because in this carbon is attached to 5 atoms.
(ii) Intermediate (c) is unstable because carbon atom is sp2 hybridised.
(iii) Intermediate (c) is stable because carbon atom is sp2 hybridised.
(iv) Intermediate (c) is less stable than the reactant (b).
उत्तर
(i) Intermediate (c) is unstable because in this carbon is attached to 5 atoms.
(iv) Intermediate (c) is less stable than the reactant (b).
Explanation:
In the transition state, the carbon atom is simultaneously bonded to incoming nucleophiles and the outgoing leaving group and such are unstable and cannot be isolated. This is because the carbon atom in the transition state is simultaneously bonded to five atoms and therefore is unstable.
APPEARS IN
संबंधित प्रश्न
How will you bring about the following conversion?
Bromomethane to propanone
How will you bring about the following conversion?
1-Chlorobutane to n-octane
How the following conversion can be carried out?
Chloroethane to butane
Which one of the following produces acyl halide by treatment with PCl5?
What would be the reactant and reagent used to obtain 2, 4-dimethyl pentan-3-ol?
Identify the following named reaction:
\[\ce{C2H5Br ->[Na/Dry ether] C2H5 - C2H5}\]
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
