Advertisements
Advertisements
Question
Which of the following statements are correct about the reaction intermediate?
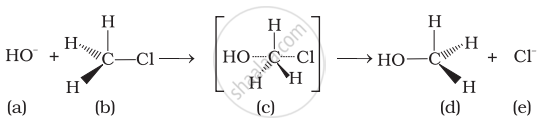
(i) Intermediate (c) is unstable because in this carbon is attached to 5 atoms.
(ii) Intermediate (c) is unstable because carbon atom is sp2 hybridised.
(iii) Intermediate (c) is stable because carbon atom is sp2 hybridised.
(iv) Intermediate (c) is less stable than the reactant (b).
Solution
(i) Intermediate (c) is unstable because in this carbon is attached to 5 atoms.
(iv) Intermediate (c) is less stable than the reactant (b).
Explanation:
In the transition state, the carbon atom is simultaneously bonded to incoming nucleophiles and the outgoing leaving group and such are unstable and cannot be isolated. This is because the carbon atom in the transition state is simultaneously bonded to five atoms and therefore is unstable.
APPEARS IN
RELATED QUESTIONS
Identify A, B, C, D, E, R and R1 in the following:

How will you bring about the following conversion?
1-Chlorobutane to n-octane
What happens when bromobenzene is treated with Mg in the presence of dry ether?
A Grignard reagent may be made by reacting magnesium with ____________.
Identify the following named reaction:
\[\ce{C2H5Br ->[Na/Dry ether] C2H5 - C2H5}\]
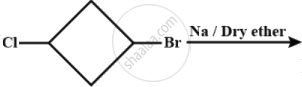 A product (1) of above reaction is:-
A product (1) of above reaction is:-
Grignard reagent shows addition on
Explain why Grignard reagents should be prepared under anhydrous conditions.
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
