Advertisements
Advertisements
प्रश्न
Which of the following statements are correct about the mechanism of this reaction?
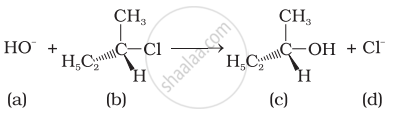
(i) A carbocation will be formed as an intermediate in the reaction.
(ii) \[\ce{OH-}\] will attach the substrate (b) from one side and \[\ce{Cl-}\] will leave it simultaneously from other side.
(iii) An unstable intermediate will be formed in which \[\ce{OH-}\] and \[\ce{Cl-}\] will be attached by weak bonds.
(iv) Reaction proceeds through SN1 mechanism.
उत्तर
(i) A carbocation will be formed as an intermediate in the reaction.
(iv) Reaction proceeds through SN1 mechanism.
Explanation:
It occurs in two steps. In step I, the polarised \[\ce{C - Cl}\] bond undergoes slow cleavage to produce carbonation and a chloride ion. The carbonation thus formed is then attacked by nucleophiles in step II to complete the substitution reaction. It is noteworthy that the 2° halides may proceed either through SN1 or SN2 type. The preservation of the integrity of the spatial arrangement of bonds to an asymmetric centre during a chemical reaction depit that the reaction is followed by SN1 mechanism.
