Advertisements
Advertisements
प्रश्न
Account for the following:
Mn shows the highest oxidation state of +7 with oxygen but with fluorine, it shows oxidation state of +4.
Give reasons:
Mn shows the highest oxidation state of +7 with oxygen but with fluorine, it shows the highest oxidation state of +4.
उत्तर १
In case of oxygen, Mn shows the highest oxidation state of +7. This is because Mn forms pπ–dπ multiple bonds using 2p orbitals of oxygen and 3d orbitals of Mn. With F, Mn displays an oxidation state of +4 because of the single bond formation caused by the unavailability of 2p orbitals in F for multiple bonding.
उत्तर २
Mn shows the highest oxidation state of +7 with oxygen because it can form ppi−dpi multiple bonds using 2p orbital of oxygen and 3d orbital of Mn. On the other hand, Mn shows the highest oxidation state of +4 with flourine because it can form single bond only.
APPEARS IN
संबंधित प्रश्न
What are interstitial compounds?
The `E_((M^(2+)//M))^Θ` value for copper is positive (+0.34 V). What is possibly the reason for this? (Hint: consider its high ΔaHΘ and low ΔhydHΘ)
What is meant by 'disproportionation'? Give two examples of disproportionation reaction in aqueous solution.
Write balanced chemical equations for the conversion of `CrO_4^(2-)` to `Cr_2O_7^(2-)` in acidic medium and `Cr_2O_7^(2-)` to `CrO_4^(2-)`
in basic medium.
Maximum magnetic moment is shown by ____________.
EΘ of Cu is + 0.34V while that of Zn is – 0.76V. Explain.
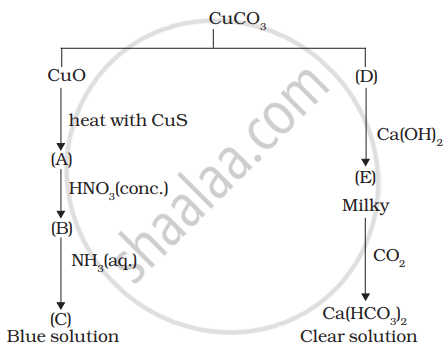
Identify A to E and also explain the reactions involved.
Assertion (A): Transition metals have high enthalpy of atomisation.
Reason (R): Greater number of unpaired electrons in transition metals results in weak metallic bonding.
Explain the magnetic properties of d-block (or transition) elements.
Account for the following:
Zirconium (Zr) and Hafnium (Hf) are difficult to separate.
