Advertisements
Advertisements
प्रश्न
What are interstitial compounds?
What are interstitial compounds? Why do these compounds have higher melting points than corresponding pure metals?
उत्तर १
Interstitial compounds are formed when small atoms such as H, C or N are trapped inside the crystal lattices of metals. They are usually non-stoichiometric and are neither typically ionic nor covalent, for example, TiC, Mn4N, Fe3H, VH0.56 and TiH1.7, etc.
उत्तर २
- Interstitial compounds are those which are formed when small atoms like H, C, N, B etc. are trapped inside the crystal lattice of metals.
- They have a melting point higher than metals due to stronger metal-non-metal bonds or compared to metal-metal bonds in pure metals.
संबंधित प्रश्न
Explain why is Fe3+ more stable than Fe2+?
ln which pair highest oxidation states of transition metals are found:
Why do the transition elements have higher enthalpies of atomisation?
The `E_((M^(2+)//M))^Θ` value for copper is positive (+0.34 V). What is possibly the reason for this? (Hint: consider its high ΔaHΘ and low ΔhydHΘ)
How would you account for the irregular variation of ionization enthalpies (first and second) in the first series of the transition elements?
Why is the highest oxidation state of a metal exhibited in its oxide or fluoride only?
Why are Mn2+ compounds more stable than Fe2+ towards oxidation to their +3 state?
Explain briefly how +2 state becomes more and more stable in the first half of the first row transition elements with increasing atomic number?
Which of the d-block elements may not be regarded as the transition elements?
How is the variability in oxidation states of transition metals different from that of the non-transition metals? Illustrate with examples.
What are alloys?
Complete and balance the following chemical equations
`MnO_4^(-) + H_2O + I^(-) ->`
Why do transition metal ions possess a great tendency to form complexes?
Why do transition elements show variable oxidation states ? In 3d series (Sc to Zn), which elements shows the maximum number of oxidation state and why ?
Account for the following :
Ti4+ is colourless whereas V4+ is coloured in an aqueous solutions.
Give reasons for the following:
The transition metals generally form coloured compounds.
Maximum magnetic moment is shown by ____________.
In lake test for Al3+ ions, there is the formation of coloured ‘floating lake’. It is due to ______.
When acidified \[\ce{K2Cr2O7}\] solution is added to \[\ce{Sn^{2+}}\] salts then \[\ce{Sn^{2+}}\] changes to ______.
Transition elements form binary compounds with halogens. Which of the following elements will form \[\ce{MF3}\] type compounds?
(i) \[\ce{Cr}\]
(ii) \[\ce{Co}\]
(iii) \[\ce{Cu}\]
(iv) \[\ce{Ni}\]
Ionisation enthalpies of Ce, Pr and Nd are higher than Th, Pa and U. Why?
Match the properties given in Column I with the metals given in Column II.
| Column I (Property) | Column II (Metal) | |
| (i) | An element which can show +8 oxidation state | (a) \[\ce{Mn}\] |
| (ii) | 3d block element that can show | (b) \[\ce{Cr}\] |
| upto +7 oxidation state | (c) \[\ce{Os}\] | |
| (iii) | 3d block element with highest melting point | (d) \[\ce{Fe}\] |
Match the solutions given in Column I and the colours given in Column II.
| Column I (Aqueous solution of salt) |
Column II (Colour) |
| (i) \[\ce{FeSO2.7H2O}\] | (a) Green |
| (ii) \[\ce{NiCl2.4H2O}\] | (b) Light pink |
| (iii) \[\ce{MnCl2.4H2O}\] | (c) Blue |
| (iv) \[\ce{CoC12,6H2O}\] | (d) Pale green |
| (v) \[\ce{Cu2 Cl2}\] | (e) Pink |
| (f) Colourless |
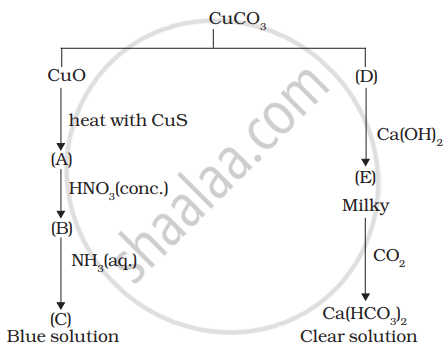
Identify A to E and also explain the reactions involved.
When a chromite ore (A) is fused with sodium carbonate in free excess of air and the product is dissolved in water, a yellow solution of compound (B) is obtained. After treatment of this yellow solution with sulphuric acid, compound (C) can be crystallised from the solution. When compound (C) is treated with KCl, orange crystals of compound (D) crystallise out. Identify A to D and also explain the reactions.
When an oxide of manganese (A) is fused with KOH in the presence of an oxidising agent and dissolved in water, it gives a dark green solution of compound (B). Compound (B) disproportionates in neutral or acidic solution to give purple compound (C). An alkaline solution of compound (C) oxidises potassium iodide solution to a compound (D) and compound (A) is also formed. Identify compounds A to D and also explain the reactions involved.
The spin magnetic moment of cobalt in the compound Hg [Co(SCN)4] is:-
Mercury is the only metal liquid at room temperature due to its:-
Agcl is soluble in NH4OH. The solubility is due to the information of:-
Which of the following transition metal is not coloured?
The number of terminal oxygen atoms present in the product B obtained from the following reactions is:
\[\ce{FeCr2O4 + Na2CO3 + O2 -> A + Fe2O3 + CO2}\]
\[\ce{A + H^+ -> B + H2O + Na^+}\]
The disproportionation of \[\ce{MnO^{2-}_4}\] in acidic medium resulted in the formation of two manganese compounds A and B. If the oxidation state of Mn in B is smaller than that of A, then the spin-only magnetic moment (µ) value of B in BM is ______. (Nearest integer)
Which of the following characteristics of transition metals is associated with their catalytic activity?
Account for the following:
Ce4+ is a strong oxidising agent.
Which one among the following metals of the 3d series has the lowest melting point?
Which of the following ions has the electronic configuration 3d6?
(Atomic number: Mn = 25, Co = 27, Ni = 28)
In order to protect iron from corrosion, which one will you prefer as a sacrificial electrode, Ni or Zn? Why? (Given standard electrode potentials of Ni, Fe and Zn are -0.25 V, -0.44 V and -0.76 V respectively.)
Give two similarities in the properties of Sc and Zn.
A pair of coloured ions is ______.
Explain the magnetic properties of d-block (or transition) elements.
Explain the use of different transition metals as catalysts.
The compounds of \[\ce{Ti^4+}\] ions are colourless due to ______.
Give a reason for the following.
Some transition metals and their compounds get attracted towards the magnetic field.
Decide which of the following atomic numbers are the atomic numbers of the inner transition elements:
29, 59, 74, 95, 102, 104
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Ionisation enthalpies
