Advertisements
Advertisements
प्रश्न
Given reasons: The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
उत्तर
Reasons behind the dipole moment of chlorobenzene being lower than that of cyclohexyl chloride are follows:
(a) In chlorobenzene, the C−Cl bond is shorter due to partial double bond character.
(b) Hybridisations of C in C−Cl in chlorobenzene and cyclohexyl chloride are sp2and sp3, respectively (sp2 C being more electronegative than sp3 C).
As dipole moment is the product of charge and bond distance, both the factors in chlorobenzene are lower as compared to cyclohexyl chloride. Therefore, the dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
संबंधित प्रश्न
Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
Which of the following reactions is an example of nucleophilic substitution reaction?
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
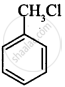
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
Which reagent will you use for the following reaction?
\[\ce{CH3CH2CH2CH3 -> CH3CH2CH2CH2Cl + CH3CH2CHClCH3}\]
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Which of the following is the definition of chirality?
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
