Advertisements
Advertisements
प्रश्न
Given reasons: The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
उत्तर
Reasons behind the dipole moment of chlorobenzene being lower than that of cyclohexyl chloride are follows:
(a) In chlorobenzene, the C−Cl bond is shorter due to partial double bond character.
(b) Hybridisations of C in C−Cl in chlorobenzene and cyclohexyl chloride are sp2and sp3, respectively (sp2 C being more electronegative than sp3 C).
As dipole moment is the product of charge and bond distance, both the factors in chlorobenzene are lower as compared to cyclohexyl chloride. Therefore, the dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
संबंधित प्रश्न
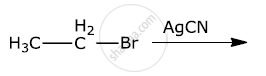
Write the major products(s) in the following:
Which would undergo SN1 reaction faster in the following pair and why?
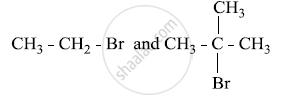
What happens when ethyl chloride is treated with aqueous KOH?
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
What is the action of the following on ethyl bromide:
moist silver oxide
Isopropyl chloride undergoes hydrolysis by:
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Which of the following statements are correct about this reaction?
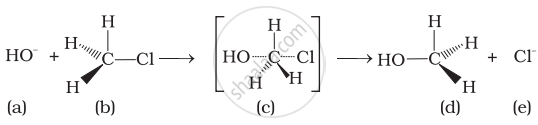
(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
In SN1 reactions, the correct order of reactivity for the following compounds:
CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is ______.
Retention of configuration is observed in ______.
