Advertisements
Advertisements
प्रश्न
What happens when ethyl chloride is treated with aqueous KOH?
उत्तर १
\[\ce{CH3 - CH2 - Cl ->[KOH/H2O]\underset{(S_N2 reaction)}{CH3 - CH2 - OH}}\]
उत्तर २
When ethyl chloride is treated with aqueous KOH, it undergoes dehydrohalogenation, which eliminates hydrogen and halogen atoms from nearby carbon atoms, resulting in the production of an alkene.
\[\ce{\underset{Ethyl chloride}{CH3 - CH2 - Cl} + KOH (aq) ->[hydrolysis][\Delta] \underset{Ethyl alcohol}{CH3 - CH2 - OH} + KCl}\]
संबंधित प्रश्न
Out of 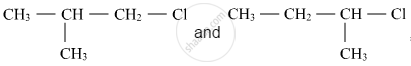 , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Write the structure of the major product in each of the following reaction :
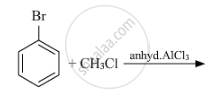
Write the mechanism of the following reaction:
\[\ce{{n}BuBr + KCN ->[EtOH-H2O] {n}BuCN}\]
What happens when chlorobenzene is subjected to hydrolysis?
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Which of the following is an example of SN2 reaction?
In the SN1 reaction, racemization takes place. It is due to:
Isopropyl chloride undergoes hydrolysis by:
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
Which of the following is an optically active compound?
SN1 reaction of alkyl halides lead to ___________.
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
A primary alkyl halide would prefer to undergo ______.
Which of the following statements are correct about this reaction?
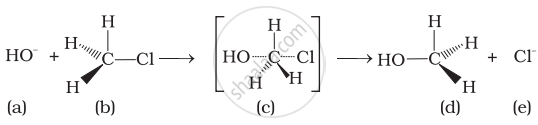
(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
CCl4 is insoluble in water because:-
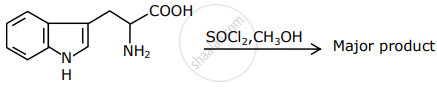
The major product formed in the following reaction is: