Advertisements
Advertisements
प्रश्न
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
उत्तर
\[\ce{C6H5-CH2-Cl}\]
SN1 reaction proceeds through the formation of carbonation intermediate.
\[\ce{C6H5CH2Cl}\] readily undergoes ionization to give \[\ce{C6H5CH^{+}2}\] carbocation, which is stabilized by resonance.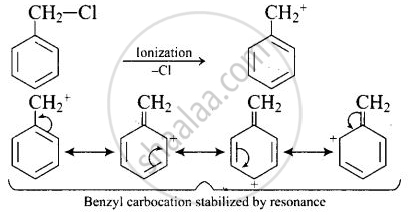
On the other hand, \[\ce{CH3CH2Cl}\] does not undergo ionization to give \[\ce{CH3CH^{+}2}\] carbonation. Therefore, \[\ce{C6H5CH2Cl}\] reacts faster than \[\ce{CH3CH2Cl}\] with \[\ce{OH-}\] ion.
APPEARS IN
संबंधित प्रश्न
Which compound in the following pair will react faster in SN2 reaction with OH−?
(CH3)3CCl or CH3Cl
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
Given reasons: The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Which of the following is an example of SN2 reaction?
Which one is most reactive towards SN1 reaction?
Most reactive halide towards SN1 reaction is ____________.
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Which of the following compounds will show retention in configuration on nucleophile substitution by OH− ion?
