Advertisements
Advertisements
Question
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Solution
\[\ce{C6H5-CH2-Cl}\]
SN1 reaction proceeds through the formation of carbonation intermediate.
\[\ce{C6H5CH2Cl}\] readily undergoes ionization to give \[\ce{C6H5CH^{+}2}\] carbocation, which is stabilized by resonance.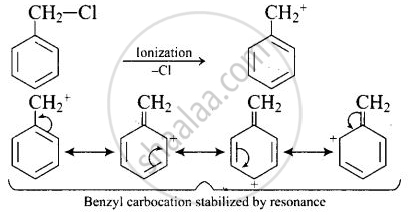
On the other hand, \[\ce{CH3CH2Cl}\] does not undergo ionization to give \[\ce{CH3CH^{+}2}\] carbonation. Therefore, \[\ce{C6H5CH2Cl}\] reacts faster than \[\ce{CH3CH2Cl}\] with \[\ce{OH-}\] ion.
APPEARS IN
RELATED QUESTIONS
Write the structures of A, B and C in the following:

What are ambident nucleophiles? Explain with an example.
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH.
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Halogenation of alkanes is ____________.
Racemic compound has ____________.
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.
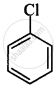
(I)
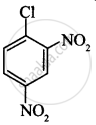
(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
A primary alkyl halide would prefer to undergo ______.
Which of the following statements are correct about the kinetics of this reaction?
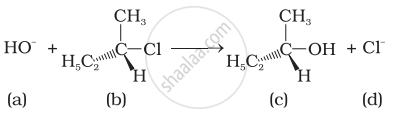
(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
