Advertisements
Advertisements
प्रश्न
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
उत्तर
\[\ce{C6H5-CH2-Cl}\]
SN1 reaction proceeds through the formation of carbonation intermediate.
\[\ce{C6H5CH2Cl}\] readily undergoes ionization to give \[\ce{C6H5CH^{+}2}\] carbocation, which is stabilized by resonance.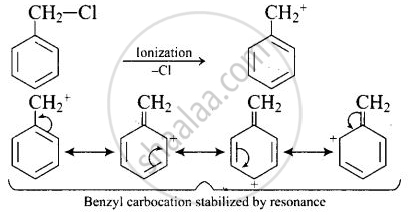
On the other hand, \[\ce{CH3CH2Cl}\] does not undergo ionization to give \[\ce{CH3CH^{+}2}\] carbonation. Therefore, \[\ce{C6H5CH2Cl}\] reacts faster than \[\ce{CH3CH2Cl}\] with \[\ce{OH-}\] ion.
APPEARS IN
संबंधित प्रश्न
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?
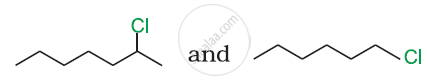
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
Which of the following reactions is an example of nucleophilic substitution reaction?
Which one is most reactive towards SN1 reaction?
Racemic compound has ____________.
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Which of the following alkyl halides will undergo SN1 reaction most readily?
CCl4 is insoluble in water because:-
Racemisation occurs in ______.
Discuss SN2 mechanism of methyl bromide using aqueous KOH.
