Advertisements
Advertisements
प्रश्न
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
Advertisements
उत्तर
The SN2 reaction involves the formation of a transition state with the carbon atom surrounded by 5 additional atoms (groups). A transition state requires minimum steric interactions. The most suitable substrates for SN2 reactions are 1° alkyl halides, followed by 2° and 3° alkyl halides. The order of reactivity towards SN2 is 1° > 2° > 3°> aryl halide. Based on this, the order will be
\[\begin{array}{cc}
\ce{CH3}\phantom{...............................}\ce{CH3}\phantom{.................}\ce{CH3}\phantom{.....}\\
\phantom{.....}|\phantom{...................................}|\phantom{....................}|\phantom{............}\\
\ce{\underset{1-Bromo-3-methylbutane}{H3C - CH - CH2 - CH2Br} > \ce{CH3 - CH - CH - CH3} > \ce{CH3 - C - CH2 - CH3}}\\
\phantom{.......................}|\phantom{..........................}|\\
\phantom{.........................}\ce{\underset{2-Bromo-3-methylbutane}{Br}\phantom{.........}\ce{\underset{2-Bromo-2-methylbutane}{Br}}}\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Write the structures of A, B and C in the following:

In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?
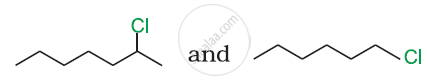
Which compound in the following pair will react faster in SN2 reaction with OH−?
CH3Br or CH3I
What happens when chlorobenzene is subjected to hydrolysis?
What is the action of the following on ethyl bromide?
silver acetate
Which of the following is optically inactive?
Which one is most reactive towards SN1 reaction?
Which among MeX, RCH2X, R2CHX and R3CX is most reactive towards SN2 reaction?
Which of the following is the correct order of decreasing SN2 reactivity?
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) | 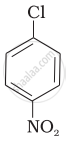 |
| (II) | 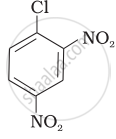 |
| (III) | 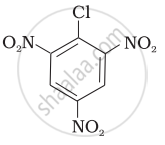 |
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
Chlorination of alkanes is an example of
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
Convert bromoethane to propanamine.
