Advertisements
Advertisements
प्रश्न
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
उत्तर
SN2 reaction involves the formation of transition state. Therefore, reactivity towards SN2 depends upon the steric hindrance:
Due to the presence of 2 methyl group in 2-Bromo-2-methylbutane it is least reactive towards SN2 reactions, 2-bromopentane is moderately reactive while 1-bromopentane is most reactive.
2-Bromo-2-methylbutane < 2-Bromopentane < 1-Bromopentane
APPEARS IN
संबंधित प्रश्न
Write the main products when methyl chloride is treated with AgCN.
Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
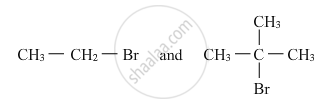
Which would undergo SN2 reaction faster in the following pair and why ?
What are ambident nucleophiles? Explain with an example.
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
What happens when ethyl chloride is treated with aqueous KOH?
What is the action of the following on ethyl bromide:
moist silver oxide
What is the action of the following on ethyl bromide?
moist silver oxide
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
