Advertisements
Advertisements
Question
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
Advertisements
Solution
The SN2 reaction involves the formation of a transition state with the carbon atom surrounded by 5 additional atoms (groups). A transition state requires minimum steric interactions. The most suitable substrates for SN2 reactions are 1° alkyl halides, followed by 2° and 3° alkyl halides. The order of reactivity towards SN2 is 1° > 2° > 3°> aryl halide. Based on this, the order will be
\[\begin{array}{cc}
\ce{CH3}\phantom{...............................}\ce{CH3}\phantom{.................}\ce{CH3}\phantom{.....}\\
\phantom{.....}|\phantom{...................................}|\phantom{....................}|\phantom{............}\\
\ce{\underset{1-Bromo-3-methylbutane}{H3C - CH - CH2 - CH2Br} > \ce{CH3 - CH - CH - CH3} > \ce{CH3 - C - CH2 - CH3}}\\
\phantom{.......................}|\phantom{..........................}|\\
\phantom{.........................}\ce{\underset{2-Bromo-3-methylbutane}{Br}\phantom{.........}\ce{\underset{2-Bromo-2-methylbutane}{Br}}}\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the major products(s) in the following:
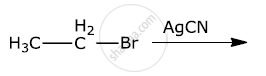
How do you convert the following:
Ethanol to propanenitrile
Write the main products when methyl chloride is treated with AgCN.
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
CH3CH2CH2CH2Br or \[\begin{array}{cc}
\ce{CH3CH2CHCH3}\\
\phantom{...}|\\
\phantom{....}\ce{Br}\
\end{array}\]
Which compound in the following pair will react faster in SN2 reaction with OH−?
(CH3)3CCl or CH3Cl
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
Identify 'A' in the following reaction -
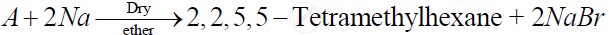
(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
Most reactive halide towards SN1 reaction is ____________.
Optically active isomers but not mirror images are called ____________.
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.
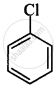
(I)
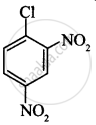
(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
The reaction of C6H5–CH=CH–CH3 with HBr produces:
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Complete the following analogy:
Same molecular formula but different structures: A : : Non superimposable mirror images: B
A primary alkyl halide would prefer to undergo ______.
Which of the following statements are correct about this reaction?
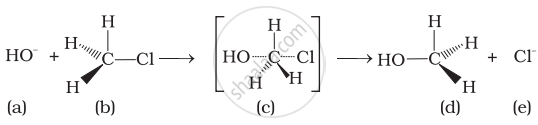
(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Chlorination of alkanes is an example of
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CH2CHCH3}\\\phantom{...}|\\\phantom{....}\ce{Br}\end{array}\] or \[\begin{array}{cc}\phantom{.....}\ce{CH3}\\\phantom{..}|\\\ce{H3C - C - Br}\\\phantom{..}|\\\phantom{....}\ce{CH3}\end{array}\]
