Advertisements
Advertisements
Question
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
Advertisements
Solution
The SN2 reaction involves the formation of a transition state with the carbon atom surrounded by 5 additional atoms (groups). A transition state requires minimum steric interactions. The most suitable substrates for SN2 reactions are 1° alkyl halides, followed by 2° and 3° alkyl halides. The order of reactivity towards SN2 is 1° > 2° > 3°> aryl halide. Based on this, the order will be
\[\begin{array}{cc}
\phantom{..........................................................}\ce{CH3}\phantom{..................}\ce{CH3}\\
\phantom{........................................................}|\phantom{......................}|\\
\ce{\underset{1-Bromobutane}{CH3(CH2)CH2Br} > \ce{\underset{1-Bromo-3-methylbutane}{(CH3)2 - CH - CH2 - CH2Br} > \ce{\underset{1-Bromo-2-methylbutane}{CH3 - CH2 - CH - CH2Br} > \ce{CH3 - C - CH2Br}}}}\\
\phantom{...............................................................................}|\\
\phantom{...................................................................................}\ce{\underset{1-Bromo-2, 2-dimethylpropane}{CH3}}\
\end{array}\]
Although all alkyl halides are 1°, the order of reactivity depends on the steric barrier around the carbon bearing the -Br atom. The more bulky groups around a carbon, the lower its reactivity towards SN2.
APPEARS IN
RELATED QUESTIONS
Write the main products when methyl chloride is treated with AgCN.
How will you bring about the following conversion?
Toluene to benzyl alcohol
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
Which would undergo SN2 reaction faster in the following pair and why ?
CH3 – CH2 – Br and CH3 – CH2 – I
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
What is the action of the following on ethyl bromide?
moist silver oxide
Optically active isomers but not mirror images are called ____________.
SN1 reaction of alkyl halides lead to ___________.
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
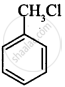
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
Which of the following statements are correct about the kinetics of this reaction?
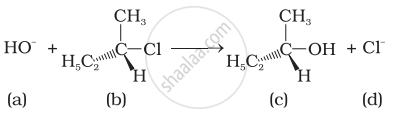
(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Which of the following is the definition of chirality?
Which one of the following compounds is more reactive towards SN1 reaction?
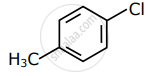
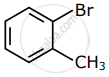
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
 |
 |
 |
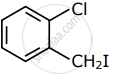 |
| I | II | III | IV |
Retention of configuration is observed in ______.
Inversion of configuration occurs in ______.
Complete the reaction with the main product formed:

Discuss the mechanism of alkaline hydrolysis of methyl bromide.
