Advertisements
Advertisements
प्रश्न
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) | 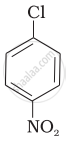 |
| (II) | 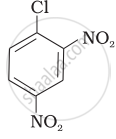 |
| (III) | 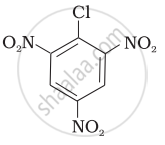 |
उत्तर
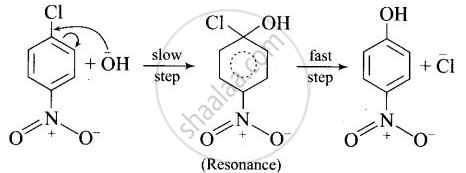
After the attachment of the nucleophile at the carbon carrying -Cl, the intermediate compound is stabilised due to resonance. Due to electron-withdrawing nature of-NO2, the nucleophile is easily attached to the benzene ring. Greater the number of -NO2 groups in the molecule, greater will be the ease with which the nucleophile will be attached. Hence, the order of reactivity is III > II > I.
APPEARS IN
संबंधित प्रश्न
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?
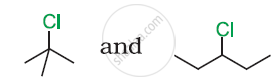
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
Which of the following is optically inactive?
Which one of the following halogen compounds is difficult to be hydrolysed by SN1 mechanism?
Optically active isomers but not mirror images are called ____________.
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Which of the following statements are correct about this reaction?
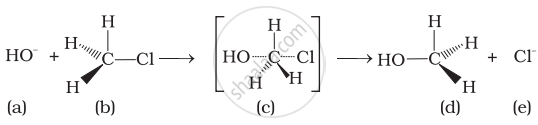
(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
