Advertisements
Advertisements
प्रश्न
Out of 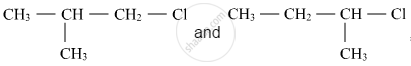 , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
उत्तर
Since formation of carbocation is the rate-determining step in the SN1 reaction, the stability of carbocation would determine its reactivity.
The order of stability of carbocation is as follows:
Tertiary > Secondary > Primary >Methyl
Here, 1-chloro-1-methylpropane would form secondary carbocation, while 1-chloro-2-methylpropane will form primary carbocation, which is less stable than secondary carbocation. Hence, reactivity towards the SN1 reaction would be higher for 1-chloro-1-methylpropane.
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
(CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
What are ambident nucleophiles? Explain with an example.
Most reactive halide towards SN1 reaction is ____________.
Optically active isomers but not mirror images are called ____________.
An important chemical method to resolve a racemic mixture makes use of the formation of ______.
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
Retention of configuration is observed in ______.
Complete the reaction with the main product formed:

