Advertisements
Advertisements
Question
What happens when ethyl chloride is treated with aqueous KOH?
Advertisements
Solution 1
\[\ce{CH3 - CH2 - Cl ->[KOH/H2O]\underset{(S_N2 reaction)}{CH3 - CH2 - OH}}\]
Solution 2
When ethyl chloride is treated with aqueous KOH, it undergoes dehydrohalogenation, which eliminates hydrogen and halogen atoms from nearby carbon atoms, resulting in the production of an alkene.
\[\ce{\underset{Ethyl chloride}{CH3 - CH2 - Cl} + KOH (aq) ->[hydrolysis][\Delta] \underset{Ethyl alcohol}{CH3 - CH2 - OH} + KCl}\]
RELATED QUESTIONS
Write the major products(s) in the following:
Out of 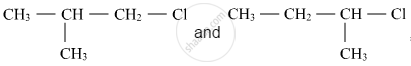 , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Write the isomers of the compound having the formula C4H9Br.
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
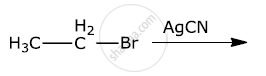
What is the action of the following on ethyl bromide?
silver acetate
AgCN reacts with haloalkanes to form isocyanide. Haloalkanes react with KCN to form alkyl cyanides as the main product. Why?
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Which of the following pairs is/are correctly matched?
| Reaction | Product | |
| I | RX + AgCN | RNC |
| II | RX + KCN | RCN |
| III | RX + KNO2 | \[\begin{array}{cc} \phantom{.......}\ce{O}\\ \phantom{.....}/\\ \ce{R - N}\phantom{....}\\ \phantom{.....}\backslash\backslash\\ \phantom{.......}\ce{O} \end{array}\] |
| IV | RX + AgNO2 | \[\ce{R-O-N=O}\] |
Which of the following is a primary halide?
Which one is most reactive towards SN1 reaction?
Which of the following is an optically active compound?
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
SN1 reaction of alkyl halides lead to ___________.
The reaction of C6H5–CH=CH–CH3 with HBr produces:
Which reagent will you use for the following reaction?
\[\ce{CH3CH2CH2CH3 -> CH3CH2CH2CH2Cl + CH3CH2CHClCH3}\]
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
Which one of the following compounds is more reactive towards SN1 reaction?
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
