Advertisements
Advertisements
Question
Which one of the following compounds is more reactive towards SN1 reaction?
Options
CH2 = CHCH2Br
C6H5CH2Br
C6H5CH (C6H5)Br
C6H5CH(CH3) Br
Solution
C6H5CH (C6H5)Br
APPEARS IN
RELATED QUESTIONS
Which would undergo SN2 reaction faster in the following pair and why ?
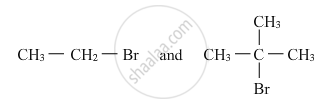
SN1 reactions are accompanied by racemization in optically active alkyl halides.
SN2 mechanism proceeds through intervention of ____________.
Among the following, the dissociation constant is highest for:
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
The reaction of C6H5–CH=CH–CH3 with HBr produces:
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Which of the following is the definition of chirality?
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
