Advertisements
Advertisements
Question
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
Solution
\[\begin{array}{cc}\ce{C2H5 - CH - CH3}\\
|\phantom{.}\\\ce{Br}\end{array}\]
Mechanism:
\[\begin{array}{cc}
\ce{H}\phantom{.................}\ce{H}\\
|\phantom{..................}|\\
\ce{H3C - C - C2H5 ->[Slow step] C^⊕ + B\overset{⊖}{r}}\\
\phantom{...}|\phantom{................}/\phantom{...}\backslash\\
\phantom{.......}\ce{Br}\phantom{...........}\ce{H3C}\phantom{....}\ce{C2H5}
\end{array}\]
\[\begin{array}{cc}
\ce{H}\phantom{....................}\ce{H}\phantom{...............}\ce{H}\phantom{.......}\\
|\phantom{.....................}|\phantom{................}|\phantom{.......}\\
\ce{C^⊕ + O\overset{⊖}{H} ->[Fast] H3C - C - OH + HO - C - CH3}\\
/\phantom{..}\backslash\phantom{....................}|\phantom{.................}|\phantom{.........}\\
\ce{H3C}\phantom{...}\ce{C2H5}\phantom{...............}\ce{C2H5}\phantom{.............}\ce{C2H5}\phantom{........}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq.ethanol]}\]
Identify 'A' in the following reaction -
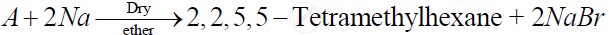
(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Halogenation of alkanes is ____________.
Optically active isomers but not mirror images are called ____________.
Racemic compound has ____________.
Among the following, the dissociation constant is highest for:
The increasing order of nucleophilicity would be:
The number of chiral carbons present in the molecule given below is ______.