Advertisements
Advertisements
प्रश्न
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
उत्तर
\[\begin{array}{cc}\ce{C2H5 - CH - CH3}\\
|\phantom{.}\\\ce{Br}\end{array}\]
Mechanism:
\[\begin{array}{cc}
\ce{H}\phantom{.................}\ce{H}\\
|\phantom{..................}|\\
\ce{H3C - C - C2H5 ->[Slow step] C^⊕ + B\overset{⊖}{r}}\\
\phantom{...}|\phantom{................}/\phantom{...}\backslash\\
\phantom{.......}\ce{Br}\phantom{...........}\ce{H3C}\phantom{....}\ce{C2H5}
\end{array}\]
\[\begin{array}{cc}
\ce{H}\phantom{....................}\ce{H}\phantom{...............}\ce{H}\phantom{.......}\\
|\phantom{.....................}|\phantom{................}|\phantom{.......}\\
\ce{C^⊕ + O\overset{⊖}{H} ->[Fast] H3C - C - OH + HO - C - CH3}\\
/\phantom{..}\backslash\phantom{....................}|\phantom{.................}|\phantom{.........}\\
\ce{H3C}\phantom{...}\ce{C2H5}\phantom{...............}\ce{C2H5}\phantom{.............}\ce{C2H5}\phantom{........}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
Identify 'A' in the following reaction -
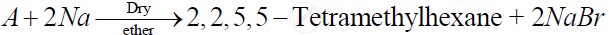
(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Which of the following is an optically active compound?
A primary alkyl halide would prefer to undergo ______.
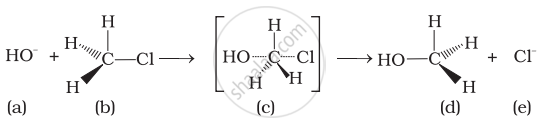
Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
How do polar solvents help in the first step in SN1 mechanism?
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
The major product formed in the following reaction is:
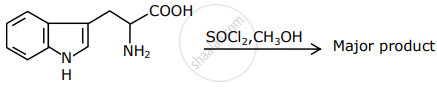
Complete the reaction with the main product formed:

