Advertisements
Advertisements
प्रश्न
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
उत्तर
\[\begin{array}{cc}\ce{C2H5 - CH - CH3}\\
|\phantom{.}\\\ce{Br}\end{array}\]
Mechanism:
\[\begin{array}{cc}
\ce{H}\phantom{.................}\ce{H}\\
|\phantom{..................}|\\
\ce{H3C - C - C2H5 ->[Slow step] C^⊕ + B\overset{⊖}{r}}\\
\phantom{...}|\phantom{................}/\phantom{...}\backslash\\
\phantom{.......}\ce{Br}\phantom{...........}\ce{H3C}\phantom{....}\ce{C2H5}
\end{array}\]
\[\begin{array}{cc}
\ce{H}\phantom{....................}\ce{H}\phantom{...............}\ce{H}\phantom{.......}\\
|\phantom{.....................}|\phantom{................}|\phantom{.......}\\
\ce{C^⊕ + O\overset{⊖}{H} ->[Fast] H3C - C - OH + HO - C - CH3}\\
/\phantom{..}\backslash\phantom{....................}|\phantom{.................}|\phantom{.........}\\
\ce{H3C}\phantom{...}\ce{C2H5}\phantom{...............}\ce{C2H5}\phantom{.............}\ce{C2H5}\phantom{........}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
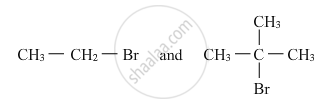
Which would undergo SN2 reaction faster in the following pair and why ?
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2OH + SOCl2 ->}\]
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH.
SN1 reactions are accompanied by racemization in optically active alkyl halides.
The process of separation of a racemic modification into d and l-enantiomers is called ____________.
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
Inversion of configuration occurs in ______.
