Advertisements
Advertisements
प्रश्न
Which would undergo SN2 reaction faster in the following pair and why ?
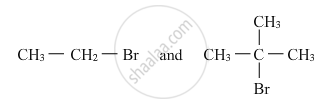
उत्तर
Primary alkyl halides prefer to undergo SN2 reactions than tertiary alkyl halides because of less steric hindrance experienced by the approaching nucleophile. Hence, out of the given pair (CH3−CH2−Br) would undergo SN2 reaction faster.
APPEARS IN
संबंधित प्रश्न
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2OH + SOCl2 ->}\]
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH.
Identify 'A' in the following reaction -
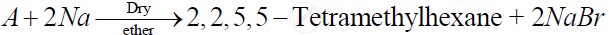
(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
Which one of the following halogen compounds is difficult to be hydrolysed by SN1 mechanism?
In the SN1 reaction, racemization takes place. It is due to:
A primary alkyl halide would prefer to undergo ______.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) | 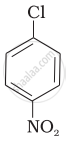 |
| (II) | 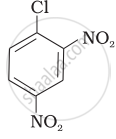 |
| (III) | 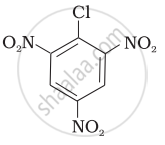 |
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
