Advertisements
Advertisements
प्रश्न
A primary alkyl halide would prefer to undergo ______.
विकल्प
SN1 reaction
SN2 reaction
α–Elimination
Racemisation
उत्तर
A primary alkyl halide would prefer to undergo SN2 reaction.
Explanation:
SN2 type reactions (i.e. bimolecular nucleophilic substitution) proceed in one step and the rate of reaction depends on concentration of alkyl halide as well as nucleophile i.e. r = k[RX][Nu]. It is a second-order reaction. During SN2 reaction, inversion in configuration occurs (viz. starting with dextrorotatory halide a laevorotatory product is obtained and vice-versa).
APPEARS IN
संबंधित प्रश्न
Write the structure of the major product in each of the following reaction :
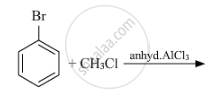
Write the structures of A, B and C in the following:

What happens when ethyl chloride is treated with aqueous KOH?
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Halogenation of alkanes is ____________.
Most reactive halide towards SN1 reaction is ____________.
Which among MeX, RCH2X, R2CHX and R3CX is most reactive towards SN2 reaction?
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.
Complete the following analogy:
Same molecular formula but different structures: A : : Non superimposable mirror images: B
