Advertisements
Advertisements
प्रश्न
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
उत्तर
In chlorobenzene, the C−Cl bond gets partial double bond character because of the +M effect shown by the Cl group. Due to double bond character, the bond length decreases in chlorobenzene as compared to the normal CH3−Cl bond length. Therefore, the C–Cl bond length in chlorobenzene is shorter than the C–Cl bond length in CH3–Cl.
APPEARS IN
संबंधित प्रश्न
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH.
Identify 'A' in the following reaction -
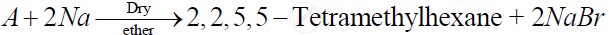
(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Among the following, the dissociation constant is highest for:
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
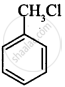
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
Which of the following statements are correct about the kinetics of this reaction?
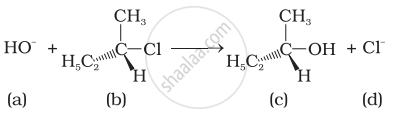
(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
How do polar solvents help in the first step in SN1 mechanism?
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CHCH2CH2Br}\\|\phantom{.........}\\\ce{CH3}\phantom{......}\end{array}\] or \[\begin{array}{cc}\ce{CH3CH2CHCH2Br}\\\phantom{}|\\\phantom{...}\ce{CH3}\end{array}\]