Advertisements
Advertisements
प्रश्न
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
उत्तर
In chlorobenzene, the C−Cl bond gets partial double bond character because of the +M effect shown by the Cl group. Due to double bond character, the bond length decreases in chlorobenzene as compared to the normal CH3−Cl bond length. Therefore, the C–Cl bond length in chlorobenzene is shorter than the C–Cl bond length in CH3–Cl.
APPEARS IN
संबंधित प्रश्न
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
What happens when ethyl chloride is treated with aqueous KOH?
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Which of the following pairs is/are correctly matched?
| Reaction | Product | |
| I | RX + AgCN | RNC |
| II | RX + KCN | RCN |
| III | RX + KNO2 | \[\begin{array}{cc} \phantom{.......}\ce{O}\\ \phantom{.....}/\\ \ce{R - N}\phantom{....}\\ \phantom{.....}\backslash\backslash\\ \phantom{.......}\ce{O} \end{array}\] |
| IV | RX + AgNO2 | \[\ce{R-O-N=O}\] |
Isopropyl chloride undergoes hydrolysis by:
An organic molecule necessarily shows optical activity if it ____________.
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
Complete the following analogy:
Same molecular formula but different structures: A : : Non superimposable mirror images: B
Which of the following statements are correct about the kinetics of this reaction?
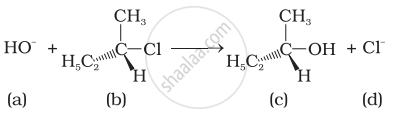
(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
