Advertisements
Advertisements
प्रश्न
Which would undergo SN1 reaction faster in the following pair and why?
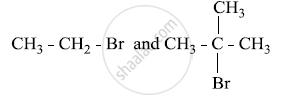
उत्तर १
Carbocations are the intermediates in the SN1 reaction. Greater the stability of the carbocations, more easily will the product be formed and hence faster will be the rate of the reaction. Because the stability of the carbocations decreases in the order: 3° carbocation > 2° carbocation > 1° carbocation > CH3+.
Therefore, the reactivity of alkyl halides towards SN1 reactions decreases in the same order, i.e. 3° alkyl halides > 2° alkyl halides > 1° alkyl halides > methyl halides.
The two structures are
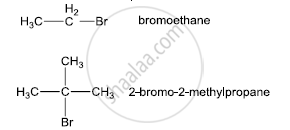
Bromoethane is a primary alkyl halide which forms a 1° carbocation intermediate in the SN1 reaction. The other compound is 2-bromo-2-methylpropane which is a tertiary alkyl halide which forms a tertiary carbocation intermediate in the SN1 reaction. Hence, 2-bromo-2-methylpropane undergoes an SN1 reaction faster than bromoethane
उत्तर २
A tertiary alkyl halide tends to undergo the SN1 mechanism because it can form a tertiary carbocation, which is stabilised by the three alkyl groups attached to it. As alkyl groups are electron donating, they allow the positive charge in the carbocation to be delocalised by the induction effect. Hence, out of the given pairs, (CH3)3C-Br would undergo SN1 reaction faster than CH3-CH2-Br.
APPEARS IN
संबंधित प्रश्न
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
CH3CH2CH2CH2Br or \[\begin{array}{cc}
\ce{CH3CH2CHCH3}\\
\phantom{...}|\\
\phantom{....}\ce{Br}\
\end{array}\]
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
SN1 reaction of alkyl halides lead to ___________.
Which reagent will you use for the following reaction?
\[\ce{CH3CH2CH2CH3 -> CH3CH2CH2CH2Cl + CH3CH2CHClCH3}\]
Which of the following statements are correct about the kinetics of this reaction?
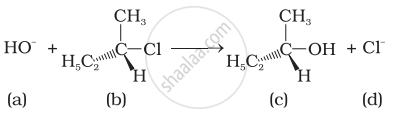
(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
Compound ‘A’ with molecular formula \[\ce{C4H9Br}\] is treated with aq. \[\ce{KOH}\] solution. The rate of this reaction depends upon the concentration of the compound ‘A’ only. When another optically active isomer ‘B’ of this compound was treated with aq. \[\ce{KOH}\] solution, the rate of reaction was found to be dependent on concentration of compound and \[\ce{KOH}\] both.
(i) Write down the structural formula of both compounds ‘A’ and ‘B’.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
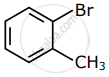
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
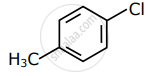 |
 |
 |
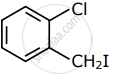 |
| I | II | III | IV |
