Advertisements
Advertisements
प्रश्न
Out of MgCl2 and AlCl3, which one is more effective in causing coagulation of negatively charged sol and why?
उत्तर
According to the Schulze-Hardy rule, the effectiveness of the salt causing flocculation depends on the charge on the ion of opposite sign to the charge on the sol particles. The greater the magnitude of the opposite charge, the higher the ability of a salt to coagulate the sol.
Thus, trivalent salt AlCl3 is more effective in causing the coagulation of a negatively charged sol than divalent salt MgCl2.
APPEARS IN
संबंधित प्रश्न
What is the reason for the stability of colloidal sols?
Out of BaCl2 and KCl, which one is more effective in causing coagulation of a negatively charged colloidal Sol? Give reason.
Give reasons for the following observations: Leather gets hardened after tanning.
What modification can you suggest in the Hardy-Schulze law?
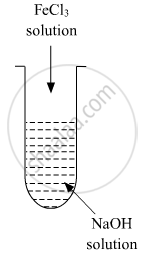
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?
Which of the following will show Tyndall effect?
The right option for the statement “Tyndall effect is exhibited by”, is ______.
Define coagulation.
Which of the following is a correct statement?
In order to cause coagulation of 100 ml Arsenious sulphide sol 111.7 mg of 2 M NaCl solution is used, the coagulation value of NaCl will be ______.
