Advertisements
Advertisements
प्रश्न
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?
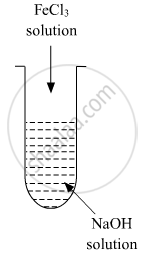
उत्तर
Colloidal particles usually adsorb those ions which are in excess and are common to its own lattice. This preferential adsorption of a particular type of ions imparts a particular type of charge to colloidal particles.
When FeCl3 is added in NaOH, the constituent of the sol is Fe(OH)3 but the dispersion medium is having an excess of OH- ions. Hence, it gets preferentially adsorbed to the sol giving the overall negative charge. The sol is represented as Fe2O3.xH2O / OH-

APPEARS IN
संबंधित प्रश्न
Define the following terms: Associated colloids
Give reasons for the following observations:
Brownian movement provides stability to the colloidal solution.
What modification can you suggest in the Hardy-Schulze law?
Explain what is observed When a beam of light is passed through a colloidal sol.
What happens when persistent dialysis of a colloidal solution is carried out?
What happens when a lyophilic sol is added to a lyophobic sol?
(i) Lyophobic sol is protected.
(ii) Lyophilic sol is protected.
(iii) Film of lyophilic sol is formed over lyophobic sol.
(iv) Film of lyophobic sol is formed over lyophilic sol.
Define coagulation.
State Hardy-Schulze rule.
Why a negatively charged sol is obtained when AgNO3 solution is added to KI solution?
In order to cause coagulation of 100 ml Arsenious sulphide sol 111.7 mg of 2 M NaCl solution is used, the coagulation value of NaCl will be ______.
