Advertisements
Advertisements
प्रश्न
State Hardy-Schulze rule.
उत्तर
Hardy-Schulze rule states that greater the valency of oppositely charged ion of the electrolyte being added, faster is coagulation. Greater the valency of flocculating ion, greater will be the power to coagulate.
संबंधित प्रश्न
Define the following terms: Associated colloids
What modification can you suggest in the Hardy-Schulze law?
Explain the terms Tyndall effect.
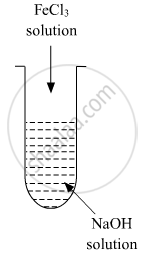
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the solution represented?

A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?
Which of the following will show Tyndall effect?
The values of colligative properties of colloidal solution are of small order in comparison to those shown by true solutions of same concentration because of colloidal particles ______.
What causes brownian motion in colloidal dispersion?
Why does leather get hardened after tanning?
Coagulation value of the electrolytes AlCl3 and NaCl for As2S3 sol are 0.093 and 52 respectively. The No. of times AlCl3 has greater coagulating power than NaCl is ______.
