Advertisements
Advertisements
प्रश्न
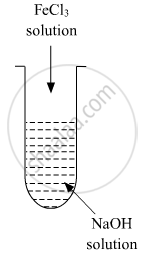
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the solution represented?

उत्तर
If FeCl3 is added to NaOH, a negatively charged sol is obtained with adsorption of OH- ions. Sol is represented as Fe2O3.xH2O/OH- (negatively charged).
APPEARS IN
संबंधित प्रश्न
Give reasons for the following observations: Leather gets hardened after tanning.
Explain the term Electrophoresis
What happens when a freshly prepared precipitate of Fe(OH)3 is shaken with a small amount of FeCl3 solution?
What happens when an emulsion is centrifuged?
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?
Match the statement given in Column I with the phenomenon given in Column II.
| Column I | Column II |
| (i) Dispersion medium moves in an electric field | (a) Osmosis |
| (ii) Solvent molecules pass through semi permeable membrane towards solvent side |
(b) Electrophoresis |
| (iii) Movement of charged colloidal particles under the influence of applied electric potential towards oppositely charged electrodes |
(c) Electroosmosis |
| (iv) Solvent molecules pass through semi permeable membranes towards solution side |
(d) Reverse osmosis |
Assertion: Colloidal solutions show colligative properties.
Reason: Colloidal particles are large in size.
Define the following term:
Coagulation
In order to cause coagulation of 100 ml Arsenious sulphide sol 111.7 mg of 2 M NaCl solution is used, the coagulation value of NaCl will be ______.
Lyophilic sols are more stable than lyophobic sols because ______.
