Advertisements
Advertisements
प्रश्न
Which one of the following has the lowest pKa value?
पर्याय
CH3 – COOH
O2N–CH2–COOH
Cl–CH2–COOH
HCOOH
MCQ
उत्तर
O2N–CH2–COOH
Explanation:
It is simpler to remove an electron since the –NO2 group is an electron-withdrawing group. As a result, the acidity is stronger, and the pKa value is lower.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
APPEARS IN
संबंधित प्रश्न
Intermolecular hydrogen bonding is strongest in ______.
Phenol reacts with Br2 in CS2 at low temperature to give ____________.
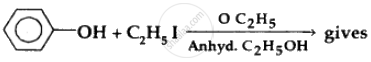
Strength of acidity is in order:
Which one of the following compounds has the most acid nature?
Which of the following statements is true:
Out of o-nitrophenol and o-cresol which is more acidic?
Phenol is used in the manufacture of
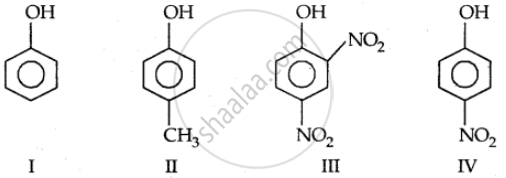
In the following compounds:
 |
 |
 |
 |
| (I) | (II) | (III) | (IV) |
The order to acidity is ______.
Compare acidity of phenol with that of ethanol.
